With the overturn of Roe v. Wade – the Supreme Court ruling protecting a pregnant woman’s liberty to choose to have an abortion – in June 2022, 44 states have since restricted abortions after a specified point or stage of pregnancy. As of April 28th, 2023, abortion is banned completely in 14 states and limited by gestational age in 5 states. In the cases of rape, incest, and fetal abnormality, abortion remains illegal in many states and will only be considered an exception when it is necessary to protect the patient’s life or health – although few exceptions are granted.
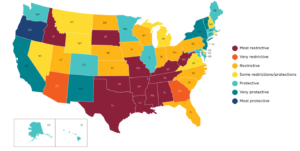
The issue of abortion is highly contentious, and with recent changes in legislation, it has become a more polarizing topic than ever before. However, one aspect that is rarely discussed in the scientific background of this debate is the definition of specific terminology used to describe the stages of pregnancy and the hallmarks of the physiological development that takes place inside a uterus. For instance, terms such as “fetus”, “embryo”, and “zygote” are often used interchangeably in popular disclosure, despite their significant biological difference. Overlooking the differences between “zygote” to “fetus”, some states have enacted laws that ban abortions after a certain gestational age (after 6, 15, 18, or 20 weeks). While the specific gestation weeks are used to signify the status of the unborn as living beings, the complex physiological development of the unborn is neglected and the possibility of fetal defects and complications is not considered. Furthermore, even the term “abortion” itself is not always clearly defined. In medical terms, an abortion can refer to any procedure that ends a pregnancy, including those performed to save the life of the mother or in cases of fetal abnormalities. However, in political disclosure, the term is often used to refer only to an elective procedure performed for personal reasons. This article is aimed to provide a clear understanding of the specific terminology and scientific background behind such a heated issue.
It should be noted that within the article, the hallmark of intrauterine development is discussed in weeks after fertilization instead of the commonly used gestational weeks. Fertilization is the union of a sperm and an egg that typically occurs about two weeks after the woman’s last menstrual period. However, the length of pregnancy in weeks of gestation is counted from the first day since the woman’s last menstrual period — two weeks before conception. Gestation weeks are used by healthcare providers because it can be difficult to pinpoint the exact date of fertilization while the end date of a woman’s menstrual period is often more easily determined. However, for the purpose of scientific discussion, using weeks after fertilization provides a more accurate measure of fetal development and allows for more precise timing when discussing medical procedures or developmental milestones. This is especially important when discussing abortions, as different laws and regulations often use gestational age as a cutoff point for when an abortion can be performed.
Zygote, Embryo, and Fetus
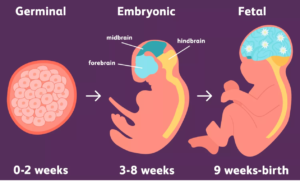
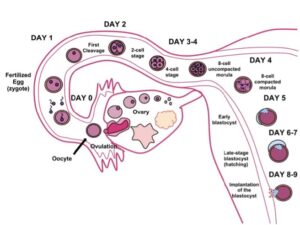
Germinal stage (weeks 0-2 / gestation week 3-4) The development of an embryo begins at fertilization when a sperm cell fertilizes an egg cell in one of the fallopian tubes to form a zygote. The zygote travels down the fallopian tube and undergoes ordinary cell divisions known as the cleavage stage. Each cycle of division takes about 24 hours. Around four to five days after fertilization, the zygote is subdivided into a 16-32 cell stage known as the morula due to its resemblance to a mulberry. The morula continues to develop into a blastocyst – a spherical ball of cells that consists of an inner cell mass and an outer cell mass. As the blastocyst starts to penetrate the uterine lining, the inner cell mass of the blastocyst gives rise to the pre-embryo and the fetal membranes surrounding the embryo will form from the outer cell mass.
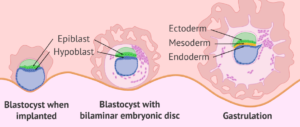
The embryonic period: embryonic development (weeks 3-5 / gestation week 5-7) The blastocyst burrowing into the uterine wall (implantation) marks the beginning of embryonic development. The embryo is the developing intrauterine organism before significant differentiation occurs. At around 17 days after fertilization, the process of gastrulation transforms the two-layered blastocyst into a multilayered and multidimensional structure known as a gastrula. As the most important step of embryonic development, the developed gastrula contains three layers – the endoderm, mesoderm, and ectoderm – that are highly functionalized and orientated for the specific concurrent development of multiple organs and organ systems. The differentiation of gastrula is tightly regulated – from the interaction between neighboring cells and cells of adjacent germ layers to the interactions at tissue boundaries coordinated through cells with dynamic morphological processes and coordinated cell fate to establish the directionality of the developing embryo.
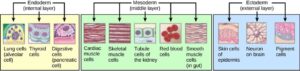
The embryonic period: organogenesis (weeks 6-8 / gestation week 8-10) The physiologically-significant organogenesis is primed by the differentiated structure of gastrula. The inner endoderm layer of the gastrula is mainly responsible for the gastrointestinal tract and respiratory tract while forming a supportive lining of the endocrine glands and urinary system. The middle mesoderm layer will develop into the cardiovascular system, reproductive system, endocrine system, lymphatic system, and connective tissues. Many internal organs such as the kidney, spleen, and ureter are also derived from the mesoderm. The outermost skin cells and pigments are formed from the upper ectoderm layer along with neurons of the brain and portions of sensory organs. As part of the embryonic period (weeks 3-8), organogenesis marks the growth and differentiation of embryonic tissues into organs.
The fetal period (weeks 9-birth / gestation week 11-birth) A fetus is a prenatal human being that has completed the embryonic stage and continues to develop and mature. By the end of week eight, primitive organ systems have developed and are prepared for further growth, maturation, and differentiation of anatomic structures throughout the entire fetal period lasting from the beginning of week nine until birth, which typically occurs around 38 weeks after fertilization. During the fetal stage, the organs and tissues that were formed continue to mature. The fetus grows in size and weight and begins to exhibit premature behaviors such as kicking and swallowing. Key developmental milestones that occur during the fetal stage include the formation of primitive fingers and toes, the development of the nervous system, and the growth of hairs and nails.
The Heart of the Matter
Development of the heart (week 3-8 / gestation week 5-10) The cardiovascular system is the first to be developed in an embryo. By the end of week three after fertilization, the passive oxygen diffusion from the maternal body is insufficient to support the metabolic demands of a developing embryo. Therefore, the development of an intrauterine fetal heart is important for the distribution of oxygen and nutrition to ensure proper embryonic and fetal development. During the third week post-conception, the flat embryonic disk starts to fold and brings the heart-forming region to its appropriate position. The folding of the embryo builds the lateral and dorsal parts of the primitive heart tube through the horseshoe-shaped cardiac crest. Different from the developed four-chambered heart, the primitive heart tube is linear and layered to give rise to the external surface, muscular bulk, and the endothelial lining of the embryonic heart. While it is a common misperception that the primitive heart tube is a segmented structure readily analogous to the developed adult human heart, it has been found that the maturation of the heart tube progresses through the gradual addition of cells. Depending on the eventual location of recruitment, the cells undergo various cellular fates to supplement the developing heat.
Via differentiation and proliferation on the outer curvature of the heat tube, the future cardiac chambers balloon. In the fourth week after fertilization, the primitive heart tube begins looping. The looping and folding of the heart tube are controlled by signaling molecules and transcriptional factors, guiding the development of the embryonic heart. The atrium and ventricular regions are formed through the expansion of the heart tube into a cardiac loop. As the heart tube continues to elongate, constrictions form between the atrial and ventricular portions that eventually give rise to the atrioventricular canal separating the atria from the ventricle. Subsequently, the right and left sides of the developing heart have separated to form the septum.
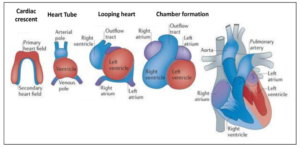
Once the primitive anatomy of the embryonic heart is developed, cardiomyocytes function as the building block of the structure to ensure the heart functions properly and accordingly. Cardiomyocytes – cells that are responsible for the contraction of the heart – are differentiated upon migrating into the inside of the heart tube, preparing to supplement the neonatal heart in an extrauterine environment after birth. During heart development, the added cardiomyocytes further differentiate from the precursor cells that make up the heart tube. All cardiomyocytes can contract, are capable of spontaneous depolarization, and are electrically coupled to neighboring cells via gap junction. Their contractility, firing frequency, and conduction velocity enables them to undergo different cell fates. Highly contractive and fast-conducting cardiomyocytes will mature the atrial and ventricular chambers due to the need for higher contraction forces. In comparison, the cardiomyocytes on the nodes of the heart are poorly electrically coupled to drive the polarization of the heart chambers.
The embryonic development begins as early as the third week after fertilization with the primary structure of a functional four-chambered heart by the end of the eighth week. By this time, the cardiac connective tissues have developed, and the atria and ventricles are fully separated from the formation of the atrioventricular valves and the septa, leaving the developed heart functionally established to pump blood effectively.
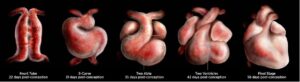
The fetal heartbeat: what is real and what is not? The famous abortion restriction legislation, the Heartbeat Bill, states that a fetal heartbeat can be detected as early as gestation week six. The detection of the claimed heartbeat is considered an indicator of life. However, as we have learned, the four-chambered heart is not developed until after eight weeks after fertilization at gestation week 10. Where is the “heartbeat” coming from?
The referred “fetal heartbeat” in legislative bans is actually the electrically-induced flickering of a portion of the fetal cardiac tissue (primitive heart tube) that will become the heart as organogenesis continues. Such cardiac activity is usually depicted with a transvaginal ultrasound around gestation week six because the probe is inserted into the vagina and positioned close to the growing embryo. Similar measures such as abdominal ultrasound can also be used to detect cardiac function at a later time in pregnancy. The ultrasound functions by sending out high-frequency waves which are reflected when it reaches different intrauterine matters such as fluids and tissue picking on the movements of the fetal cardiac tissue. The electronic sounds are translated to the sound recognizable by the human ear. As a matter of fact, the frequency of the observed sound of cardiac function is not a function of blood flowing through an artery or the strong pumping of a developed human heart as a marker of fetal personhood, but a function of primitive fetal activity for proper development.
The actual fetal cardiac activity is facilitated by the cardiomyocytes and ion channels. The conductive cardiomyocytes contract rhythmically for the formation of primitive heart tubes and the ballooning of heart chambers. The regulated contraction of the cardiomyocytes is driven by the opening and closing of ion channels. Ion channels are proteins that span the cell membrane and control the flow of charged particles such as ions in and out of the cells. As the primitive fetal heart develops and matures, ion channels allow ions to flow in and out. The moving of electrons generates the electrical signals that drive the contractions of cardiomyocytes and ultimately give rise to the formation of a chambered heart. The fetal cardiovascular system is essential to support the intrauterine metabolic demands. Each cardiac function (or heartbeat – once the heart is developed after eight weeks post-fertilization) is responsible for pumping blood throughout the developing fetal body structure and providing nutrition to the developing organs. Different from the newborn heart or the developed heart of an adult, oxygen is supplied through blood by the mother as the lung of the fetus does not function until birth. Each fetal heartbeat pushes oxygen-rich blood from the placenta through the umbilical vein into the fetal body. Once it has been distributed to the developing organs and premature tissues, the blood returns to the placenta and is replenished with fresh oxygen and nutrients. This circulation is crucial for fetal growth to ensure that the unborn develops to function properly.
Too Early, Too Late
Fetal abnormalities. The fetus continues to grow and mature from the ninth week after fertilization to birth. Bones begin to harden, reflexes begin to develop, and lungs begin to mature; yet, with greater responsibilities comes greater risk. Fetal abnormalities are unexpected or unusual conditions and developments during fetal maturation. Abnormalities are usually caused by numerous factors including fetal genetic disorders, chromosomal abnormalities, and extrauterine exposures. Because a healthy baby at birth depends on a series of important changes undergone by the developing fetus, a single genetic disorder can result in physical defects (heart defects, missing toes, spina bifida…) and cognitive disabilities (Down syndrome, fragile X syndrome, genetic conditions…), sometimes even life-threatening conditions at birth.
The emotional toll. Receiving a diagnosis of a fetal abnormality can be a traumatic and overwhelming experience, and parent(s) may experience grief, guilt, and anxiety. The process of making decisions about how to proceed with a pregnancy that has been diagnosed can be difficult and emotionally draining for parents. It is usually advised by healthcare providers to terminate the pregnancy when the fetal abnormality is severe and incompatible with life. The termination stigma, which is a deeply personal and difficult decision for the parent(s), is commonly accompanied by severe depression and postpartum psychiatric disorders.
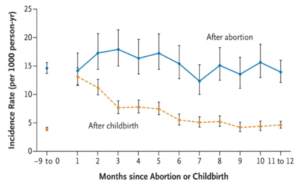
The physical toll that could’ve been avoided. Nearly 120,000 pregnancies are affected by birth defects each year and almost all fetal abnormalities can be detected through comprehensive prenatal testing. However, nearly all of the abnormalities cannot be diagnosed until well after the gestational cutoffs. Fetal abnormalities in some special areas such as the fetal head and urinary tract can be detected starting eight weeks after fertilization. However, as the fetus continues to develop from the beginning to the middle of the fetal stage at around 18 weeks after fertilization – gestation week 20 and the latest gestation limit – the detection rate of abnormalities will increase from 44% to 74%. Being forced to carry a pregnancy to term under the abortion ban, knowing that the fetus has abnormalities that are life-challenging/incompatible or life-threatening, is a traumatic experience. Putting ethical considerations aside, the process is not only emotionally challenging but physically dangerous. Some abnormalities can cause preterm (premature) labor which increases the risk of complications such as respiratory distress syndrome and cerebral palsy of the newborn.
Developmental abnormalities can cause placenta disorders that lead to bleeding and other complications, endangering both the pregnant person and the fetus. Some abnormalities also increase the risk of developing preeclampsia — a serious condition that can lead to high blood pressure and organ damage to the mother.
I Did Not Ask For It
Banning abortion with no exceptions for rape or incest, as some states have done, can lead to further trauma for the survivors of sexual violence. In the United States, there are an estimated 32,000 pregnancies resulting from rape each year in adult women. Many victims receive little to no aftercare and most experience various forms of post-traumatic stress disorder (PTSD). A third of these pregnancies are not discovered until the second trimester, and any delay in detection reduces women’s options. This is most prevalent, especially outside major urban centers, where many women are still recovering from being raped when they are called on to decide whether to have an abortion.
Regardless of legal restrictions, people will still choose to have abortions and seek out abortion services. With 18% of pregnancies ending in abortion each year, it is clear that ending a pregnancy is a common decision. Statistics have shown that there is no difference in abortion rates between countries where it is legal and those where it is not. When access to safe abortion is restricted, people may resort to unsafe, clandestine abortions, especially those who cannot afford to travel or seek private care. The criminalization of abortion does not prevent abortion; it only makes them more dangerous. Preventable deaths and injuries resulting from unsafe abortions highlight the importance of access to safe and legal abortion as a basic human right. A woman’s right to her body should be her own.
Peer editors: Mikayla Feldbauer, Raeanne Geffert, and Nila Pazhayam
SWAC Board Statement on Abortion and the Overturning of Roe v Wade