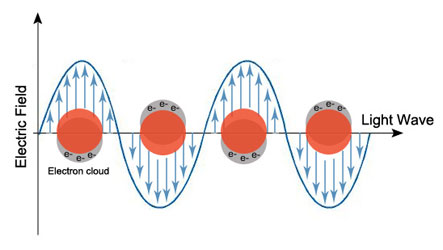
In the late winter months of 1957, Rufus Ritchie was toying around with some equations on a chalkboard at the Oak Ridge National Laboratory in Tennessee. He was attempting to clear up a phenomenon that several other researchers had noticed: something curious happened when they passed an electron beam through thin foils of different metals. After passing through the foil, the electrons had lost energy. That’s not entirely surprising– after all, we have less energy after running across a field. But the interesting part was that these electrons lost a different and, importantly, specific amount of energy depending on which metal foil they went through. That would be like having less energy after running through Bermuda grass compared to one of bluegrass, even though everything else about the field is identical. Ritchie’s explanation of this, at the time, was quite a radical one: the energy losses are due to the metal’s surface electrons interacting with the electron beam. The surface electrons would get excited by the beam and wiggle, or oscillate, back and forth at a metal-dependent frequency. Aluminum, gold, silver, and other metals all have different material properties, so they each have a different frequency. Since the electrons at the surface of a metal resemble a plasma, this frequency was dubbed a “plasma frequency,” and the oscillations dubbed “plasmons.”
Unfortunately, these results remained theoretical for a while. After all, it was the 1950’s, when scientists simply didn’t have the complex experimental equipment we do today. But Ritchie’s predictions were proven correct, and further expanded upon through the decades. Thus, the field of plasmonics was born, which has exploded into mainstream science and technology. I’ll admit that plasmonics sounds pretty abstract, but there are a few key reasons why it’s practically important.
One has to do with the way light behaves. I had a professor once say, “Light is like jelly– it doesn’t like to be squeezed.” If you try to squeeze jelly, it’ll just find a way out of your hand. If you try to focus light into too small a space, it doesn’t behave. This misbehaving can be understood by the diffraction limit, which says (loosely) that if you use a standard lens, you can’t focus light down to a space less than ½ its wavelength, mathematically represented as 𝜆/2. So if you want to focus a visible beam of, say, green light of 532 nanometers, you can’t go any smaller than 266 nanometers. But with plasmonics, you can squeeze light down further than that. If you make a nanoparticle out of a metal, it still has the characteristics that Ritchie worked out. The surface electrons oscillate no matter the size scale, so metal nanoparticles less than 200 nanometers can have plasmons that are in the visible or even infrared region of 1000 nanometers or more. The current records stand at focusing light to a space of about 𝜆/100, 50 times better than traditional lens focusing.
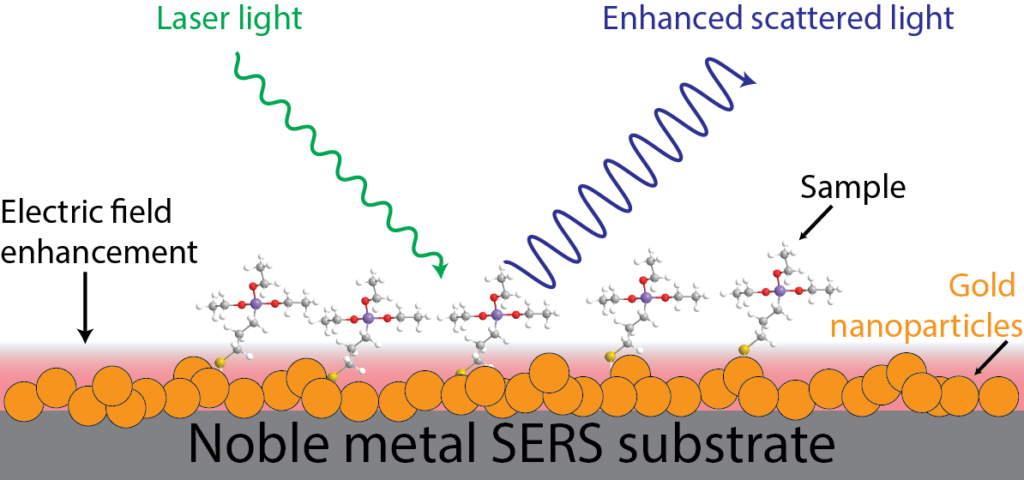
That enhanced light focusing is interesting in its own right, but it has a useful side effect: it substantially increases the electric field that surrounds the particle. That means that there is a lot more energy around the particle than normal. This increased electric field can be harnessed and utilized in different ways, such as enhancing sensing in surface enhanced Raman scattering (SERS), biosensing of certain markers of Alzheimer’s, and catalyzing chemical reactions, like the dissociation of hydrogen.
When nanoparticles support plasmons, they will often resonate around the circumference of the particle. You can picture it like a standing wave going around and around, where at each point the surface electrons are oscillating up and down. This rapid and localized oscillation generates a lot of heat, as any rapid motion would. Since the heating is only around the nanoparticle, it is extremely localized. The heating can be used to generate steam from water at room temperature; just add light! Localized plasmonic heating has also been employed in healthcare settings via techniques like photothermal therapy, which uses plasmonic nanoparticles to burn up targeted cells in the body, namely cancerous tumors (see my UNC Chemistry colleague Daniel Conroy’s 2018 article for more).
Plasmonics as a field is very much in its infancy, so with advancements in nanofabrication and technology, it seems like the sky’s the limit.
Peer Editors: Colleen Steward and Gabrielle Dardis