In 2013, cancer affected approximately 14 million people in the United States, and its direct medical costs were almost $75 billion, making cancer a devastating disease from both the human and financial perspectives. Nanotechnology may offer some relief, but at what cost?
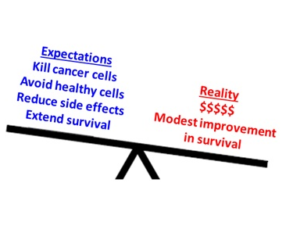
Cancer is a collection of diseases characterized by abnormal cell growth that can spread throughout the body. Chemotherapy drugs are currently the standard of care for treating many malignancies, but these anticancer drugs are not very specific for cancer cells. Conventional chemotherapeutics attack actively dividing cells, and unfortunately are unable to distinguish between cancer cells and rapidly growing healthy cells. Damage to healthy tissue leads to undesirable side effects, such as hair loss, nausea, and vomiting.
Nanotechnology scientists are interested in using nano-sized therapies to improve targeting to cancer cells. These nano-sized therapies are approximately 1/1000th the width of a strand of hair. Comparing the size of a nanoparticle to the width of human hair is like comparing the length of a sesame seed to the height of the Empire State Building.
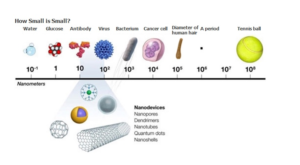
Cancer cells display unique receptors on their surface. These receptors are similar to bumper stickers on a car and serve as identifying tags so they can be recognized for imaging and therapy purposes. Small molecules, known as ligands, are designed to recognize receptors on different cancer cells. Scientists can use these ligands to coat nanoparticles containing conventional chemotherapy drugs inside to improve the delivery of drugs to tumors while avoiding healthy cells. These nanoparticles are designed with the expectation that they will selectively target and kill cancer cells, reducing adverse side effects associated with conventional chemotherapy drugs.
Although the preclinical data (from experiments performed in the laboratory on human cells and small animals) look promising, these nano-sized therapies have shown only modest improvements in extending life in cancer patients. For example, glioblastoma (GBM), the most common and aggressive brain cancer, has very poor prognosis and high recurrence rates. Without treatment, survival is about 3 months. Even with treatment, just half of GBM patients live 15 months after diagnosis. Sadly, less than 3-5% of patients survive more than five years. The poor survival rates of GBM patients are partially due to the high recurrence rates and challenges in delivering drugs to the brain. Nanoparticles can be delivered to the brain via convection-enhanced delivery (CED) or designed to cross the blood brain barrier, a protective barrier that severely limits transport of molecules to the brain; however, GBM remains incurable.
Several studies indicate nanoparticles pose a higher financial burden to patients than their unencapsulated or “free” drug counterparts. In 2009, the average cost per dose of anticancer drug doxorubicin was $62-$162 compared to $5,594 for Doxil, a nanoparticle containing doxorubicin. Similarly, the average cost per dose of anticancer drug paclitaxel was $90-$454 compared to $5,054 for Abraxane, a paclitaxel nanoformulation. Although neither nanomedicine improved patient survival, some argue that the higher cost of nanomedicines is justified because patients who received nanomedicines experienced less toxic side effects versus those who received the “free” anticancer drugs. For example, cardiotoxicity is a well-documented side effect of doxorubicin. In 2015, Cohen and Shimp wrote a letter in response to a study that reported reduced treatment costs for ovarian cancer patients undergoing Doxil treatment who received selective cardiac monitoring rather than routine cardiac monitoring. Cohen and Shimp proposed further financial savings by treating ovarian cancer patients with doxorubicin ($30/cycle) rather than Doxil ($4,363/cycle). Although doxorubicin is less expensive, more patients treated with doxorubicin experience damage to the heart compared to Doxil-treated patients.
Cancer takes a toll on its patients and their families, and unfortunately many families are forced to make an agonizing decision between extending life and accumulating debt. The concept of using nanotechnology to better treat cancers and simultaneously reduce adverse side effects is promising. However, the cost of such treatments is astronomical and the return has been minimal. This dilemma emphasizes the need to decrease research development costs of these nano-sized treatments to ultimately reduce the cost to patients.
Edited by Jet Sperlazza
Dear Christina,
thank you for posting your point a view.
In a country where people pay their treatment, the cost is really a major diference.
I would like to know when you refer to the doses prices of doxorubicin and doxil, do you consider the number of doses necessary to perform the treatment?
Kind Regards, Joana
nice job!!!!!!!!!
Thank you so much, I was looking for information like this for my chemistry assignment. It was super helpful, Good job 🙂
thank you I was working on this, this helped a lot