If you opt for a Guinness this St. Patrick’s Day, keep an eye on your pint as it settles (which, according to official Guinness standards should happen for exactly 119.5 seconds between the first pour and the top up). Fans of the Irish stout have long noticed that many of the bubbles in a fresh pint of Guinness actually seem to sink instead of floating toward the top – completely opposite from most other carbonated beverages.

Perhaps unsurprisingly, given the popularity of beer at many conferences, more than one team of scientists has set out to explore the counterintuitive bubble behavior. After one group recorded video evidence dispelling the possibility that the sinking bubbles could be an optical illusion, other groups conducted further studies, yielding two compatible explanations – the composition of the gas in the bubbles and the shape of the glass.
The bubbles in most beers are composed of carbon dioxide, which forms relatively large bubbles against the side of the glass after beer (or soda!) is depressurized and poured. Although Guinness contains some carbon dioxide, it also contains nitrogen as a key component of the gas. Since nitrogen bubbles grow slower, they end up smaller and less buoyant than their CO2 counterparts.
Bubble size alone doesn’t explain their sinking, however. Nitrogen gas is still less dense than beer, so small bubbles should still rise under ordinary circumstances. Modeling work conducted by a team from Ireland reveals how the shape of the pint glass contributes to the creation of the enigmatic sinking bubbles.
In a typical pint glass, the bottom of the glass is narrower than the top. As bubbles that formed along the bottom of the glass rise to the top, they drag some of the liquid with them up through the center, creating a bubble-rich, low-density column flowing up through the middle of the glass. However, once this upward current reaches the air-beer interface, that liquid has nowhere else to go. It circulates back down along the inside of the glass, dragging along any small Nitrogen bubbles clinging to the sides.
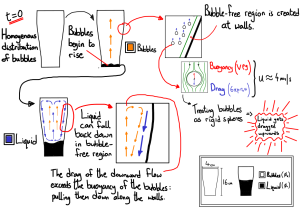
Eventually, of course, what goes down must come up (at least, if we’re talking about buoyant bubbles), and the foam collects at the top to form Guinness’s distinctive creamy, white head.
The circulation pattern should take place in any freshly-poured, wide-topped pint glass (although Guinness will tell you that their beer is best enjoyed in a branded, flared Guinness pint), and other carbonated beverages also go through this same circulation before settling. However, Guinness’s dark color and the tiny Nitrogen bubbles combine to make the cascading bubbles observable to any pub goer.
If you would like to play with these cool “fizzics” yourself this St. Patrick’s Day, you could test the importance of glass shape by opting for a graduated cylinder or inverted “anti-pint” as a drinking vessel, but for the casual fan, I recommend sticking with the traditional stout and pint combination.
Peer edited by Michelle Engle
Follow us on social media and never miss a Not So Frivolous Science post!